Type 2 diabetes mellitus is a chronic metabolic disease characterized by persistent hyperglycemia, immune dysfunction, and microvascular compromise [1]. Although diabetes is typically diagnosed and managed in medical settings, its earliest clinically significant complications may present in the oral cavity. Among these manifestations, fungal infections constitute a critical disease spectrum, ranging from common superficial mucosal involvement to rapidly progressive, life-threatening invasive infections.
Oral lesions, such as oral candidiasis, mucosal erythema, palatal ulceration, delayed epithelial healing, unexplained maxillary pain, facial paresthesia, and sinus-related symptoms in patients with known or suspected diabetes, may reflect impaired host defense mechanisms rather than isolated local pathology. Superficial fungal infections (Figure 1) often serve as early markers of immune dysregulation, whereas deep fungal infections, such as mucormycosis (Figure 2), may be catastrophic sequelae of uncontrolled disease [2,4].
Immune and Metabolic Drivers of Fungal Infection
Chronic hyperglycemia disrupts innate and adaptive immunity by impairing neutrophil and macrophage function, leading to persistent inflammation with inadequate fungal clearance, while diabetes-associated microvascular disease reduces tissue perfusion and oxygenation, compromising epithelial integrity and wound healing in oral and maxillofacial tissues and facilitating fungal invasion, with chronically elevated proinflammatory cytokines contributing to tissue damage [1]. Additionally, altered iron metabolism in hyperglycemic and acidotic states increases free iron availability, enhancing fungal growth and virulence, a mechanism especially important in mucormycosis, where iron promotes angioinvasion, thrombosis, and tissue necrosis [4,5].
Clinical Markers in Salivary Dysfunction
Salivary gland dysfunction is one of the most common oral manifestations of diabetes and plays a central role in the development of superficial fungal infections. Both subjective xerostomia and objective hyposalivation are consistently reported in individuals with type 2 diabetes. Systematic reviews estimate that approximately 40-50% of patients experience clinically significant dry mouth symptoms.
Objective studies demonstrate reduced salivary flow rates, lower salivary pH, and diminished buffering capacity in diabetic patients. Elevated salivary glucose levels further enhance fungal adherence and proliferation, particularly among Candida species. Oral candidiasis is therefore one of the most frequent fungal manifestations of diabetes, presenting as erythematous, pseudomembranous, or angular cheilitis variants [2].
Like what you’re learning? Download a brochure for our Orofacial Pain and Oral Medicine certificate or master’s degree program.
Superficial Fungal Infections
Recurrent superficial fungal infection warrants careful evaluation for systemic contributors. Persistent candidiasis despite appropriate antifungal therapy may indicate ongoing metabolic dysregulation, salivary hypofunction, or both. In some patients, superficial fungal disease represents the first clinically apparent manifestation of diabetes or impaired glucose regulation.
Superficial fungal infection should be understood along a continuum rather than as a discrete entity. Chronic mucosal colonization, epithelial breakdown, and delayed healing increase the risk of fungal penetration into deeper tissues, particularly in the presence of ischemia, neuropathy, or focal trauma.
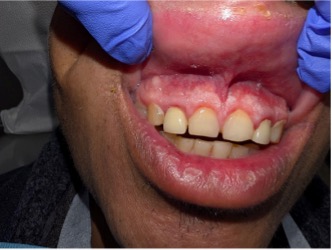
Figure 1. Superficial oral fungal infection (candidiasis) involving the anterior maxillary gingiva in uncontrolled diabetes.
Deep Fungal Infections
Beyond superficial disease, diabetes markedly increases the risk for invasive fungal infections.
Rhino-orbito-cerebral mucormycosis represents the most severe fungal complication associated with diabetes and constitutes a true medical emergency.
Early manifestations frequently involve the oral cavity and maxillofacial region. Clinicians may observe palatal ulceration or necrosis, unexplained maxillary or facial pain, tooth mobility, mucosal discoloration, paresthesia, or sinus-related symptoms. These findings often precede orbital involvement, cranial nerve deficits, and intracranial extension.
The pathogenesis of mucormycosis is characterized by aggressive angioinvasion, vascular thrombosis, and rapid tissue necrosis. Hyperglycemia, acidosis, and iron dysregulation synergistically impair host defense while enhancing fungal invasion. Even short delays in recognition and referral significantly increase morbidity and mortality [4,5].
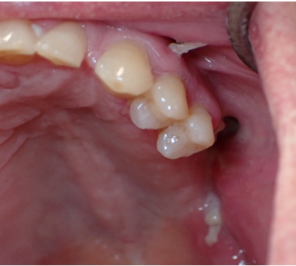
Figure 2. Invasive oral fungal infection (mucormycosis) of the left posterior maxilla in uncontrolled diabetes, demonstrating advanced tissue necrosis and Angio invasive disease.
Conclusion
The presence of recurrent candidiasis, unexplained mucosal ulceration or palatal necrosis, delayed healing, maxillary pain, facial paresthesia, or sinus-related symptoms in patients with known or suspected diabetes should prompt immediate concern for the severity and progression of fungal disease [2]. Through early recognition and communication with medical teams, oral medicine clinicians play a pivotal role in expediting diagnosis, initiating timely management, and influencing outcomes, particularly in suspected invasive fungal infections [4,5]. Preventive care, including management of xerostomia, preservation of mucosal integrity, surveillance for recurrent candidiasis, and patient counseling on hydration, glycemic control, and avoidance of mucosal trauma, supports epithelial barrier function and may limit progression from superficial to life-threatening infection. Longitudinal follow-up enables monitoring over time [1,2].
In conclusion, fungal infections constitute a critical and often under recognized oral manifestation of diabetes, ranging from superficial mucosal involvement to rapidly fatal invasive disease. Diabetes-associated immune dysfunction, microvascular compromise, altered iron metabolism, salivary hypofunction, and neuropathy collectively facilitate fungal proliferation and invasion of oral and maxillofacial tissues, underscoring the essential role of oral medicine clinicians in comprehensive, multidisciplinary diabetes care.
Are you interested in a variety of issues focused on orofacial pain, medicine and sleep disorders? Consider enrolling in the Herman Ostrow School of Dentistry of USC’s online, competency-based certificate or master’s program in Orofacial Pain and Oral Medicine.
References
[1] Kalyani, R. R., Neumiller, J. J., Maruthur, N. M., & Wexler, D. J. (2025). Diagnosis and Treatment of Type
[2] Diabetes in Adults: A Review. JAMA, 334(11), 984–1002. https://doi.org/10.1001/jama.2025.5956
[3] Lima, A. L., Illing, T., Schliemann, S., & Elsner, P. (2017). Cutaneous Manifestations of Diabetes Mellitus: A Review. American journal of clinical dermatology, 18(4), 541–553. https://doi.org/10.1007/s40257-017-0275-z
[4] Alqarihi, A., Kontoyiannis, D. P., & Ibrahim, A. S. (2023). Mucormycosis in 2023: an update on pathogenesis and management. Frontiers in cellular and infection microbiology, 13, 1254919. https://doi.org/10.3389/fcimb.2023.1254919\
[5] Steinbrink, J. M., & Miceli, M. H. (2021). Mucormycosis. Infectious disease clinics of North America, 35(2), 435–452. https://doi.org/10.1016/j.idc.2021.03.009


